Cancer research: key function of important leukaemia gene demystified
The oncogene EVI1 causes an aggressive type of leukaemia, but its exact function has been a mystery. A research team led by scientists from Vienna now showed that EVI1’s cancer causing effect relies on activating a single gene – the stem cell transcription factor ERG. The finding is reported in the journal Blood.
In cancer cells, chromosomes are often broken and reshuffled, which leads to changes in the order and regulation of genes. In our blood system, such rearrangements of chromosome 3 can activate the oncogene EVI1, which causes a particularly aggressive form of acute myeloid leukaemia (AML) that is resistant to chemotherapy and incurable in almost all cases. To search for more effective therapies, scientists are trying to understand how EVI1 causes leukaemia. However, studying cells with EVI1 rearrangements is difficult, because they grow poorly, both in culture and mice.
“We have many experimental tools to investigate gene functions in cancer, but in the case of EVI1-driven AML, we simply lacked a relevant disease model for using these tools,” says Johannes Zuber of the Research Institute of Molecular Pathology (IMP). His lab and the group of Florian Grebien at the University of Veterinary Medicine Vienna partnered in this study.
Eventually and against the odds, Inês Barbosa, then a PhD student in the Zuber lab, succeeded in creating new models of EVI1-driven AML that enabled genetic experiments. Around the same time, the team identified a human leukaemia cell line that is dependent on EVI1 – one that Zuber thinks is likely to remain an important tool for future AML research. Once these two models became available, they opened up the opportunity for a systematic investigation of the molecular pathways that involve EVI1.
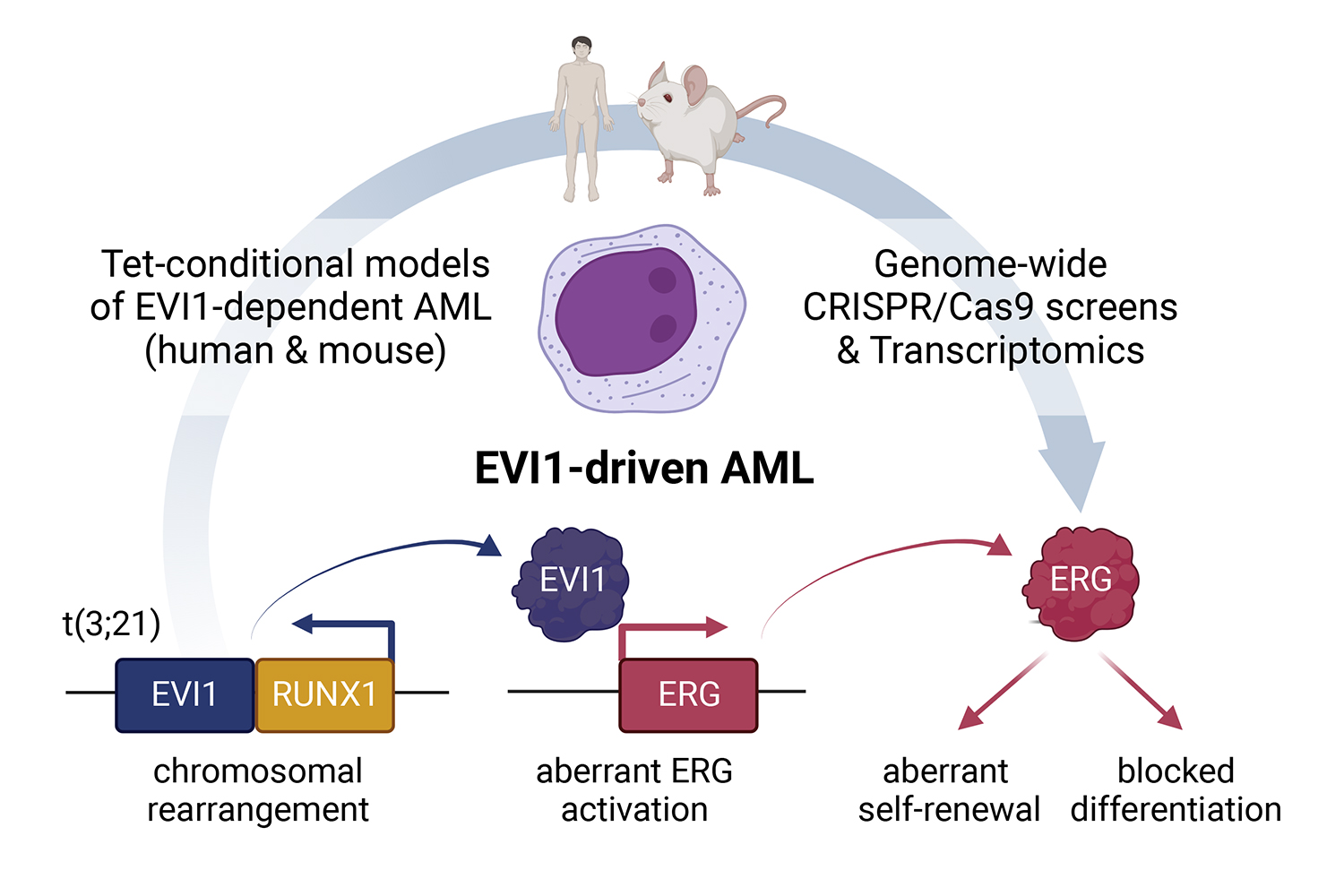
Johannes Schmoellerl, who worked as PhD student in the Grebien lab before joining the Zuber lab as a postdoc, used the “gene scissors” CRISPR-Cas9 to eliminate genes. This allowed him to screen for possible vulnerabilities of EVI1-driven leukaemia cells across the entire genome. Together, the team uncovered that the transcription factor ERG is both activated by EVI1 and required for its function in leukaemia. ERG is a known oncogene that plays a role in different forms of cancer, particularly in prostate cancer. ERG is part of a group of proteins that contribute to cell proliferation and differentiation, the development of blood vessels, and other processes. Its role in the development of EVI1-driven AML, however, had previously been unknown.
The scientists then tried to find out how important ERG really is in causing this aggressive blood cancer. By then it was clear that ERG was required for this AML to develop, but not whether other proteins were also involved. Therefore, the scientists shut down the EVI1 oncogene in cancer cells and expressed ERG artificially. As a result, the leukaemia cells survived, thereby showing that ERG alone is sufficient to compensate for loss of EVI1.
“While there are different mechanisms that can result in EVI1 expression, their main function converges on the activation of one single protein, ERG,” says Johannes Schmoellerl. “Finding this direct link between EVI1 and ERG is an important step forward,” says Florian Grebien, “Not only for understanding EVI1’s role in leukaemia, but also for our search for more effective therapies.”
Original Publication
Johannes Schmoellerl, Inês A. M. Barbosa, Martina Minnich, Florian Andersch, Leonie Smeenk, Marije Havermans, Thomas Eder, Tobias Neumann, Julian Jude, Michaela Fellner, Anja Ebert, Monika Steininger, Ruud Delwel, Florian Grebien, and Johannes Zuber:
“EVI1 drives leukemogenesis through aberrant ERG activation.” Blood, 2022, doi:https://doi.org/10.1182/blood.2022016592
About the IMP and Vienna BioCenter
The Research Institute of Molecular Pathology (IMP) in Vienna is a basic life science research institute largely sponsored by Boehringer Ingelheim. With over 200 scientists from 40 countries, the IMP is committed to scientific discovery of fundamental molecular and cellular mechanisms underlying complex biological phenomena. The IMP is part of the Vienna BioCenter, one of Europe’s most dynamic life science hubs with 2,650 people from over 80 countries in six research institutions, three universities, and 41 biotech companies.
www.imp.ac.at, www.viennabiocenter.org
About the University of Veterinary Medicine Vienna
The University of Veterinary Medicine Vienna (Vetmeduni) is one of the leading veterinary academic education and research institutions in Europe. Its main focus is on the research areas of animal health, food safety, animal husbandry and animal welfare, and basic biomedical sciences. The Vetmeduni employs 1,500 staff and currently educates 2,500 students. The campus in Vienna Floridsdorf has five university hospitals and numerous teaching and research facilities. Two research institutes on Vienna's Wilhelminenberg as well as a teaching and research estate in Lower Austria and a branch in Tyrol also belong to Vetmeduni. The Vetmeduni plays in the global top league: In the global Shanghai University Ranking 2022, it once again ranked among the top ten in the subject "Veterinary Science". www.vetmeduni.ac.at