Immunologists reveal how the Pax5 protein regulates B cell development and commitment
The IMP has a long history of research with Pax5, an essential transcription factor for B cell development: Deputy Scientific Director Meinrad Busslinger and his lab have researched its functions for decades. Sarah Grünbacher, a recent graduate from the Vienna BioCenter PhD Program, explains their latest findings on the domains of Pax5,now published in theJournal of Experimental Medicine.
Meinrad Busslinger’s lab has worked on the protein called Pax5 for a very long time, uncovering many of its functions. What does Pax5 do?
The protein Pax5 is a major regulator of B cell development. Without Pax5, no B cells and consequently, no antibody diversity can be generated. B cells originate from versatile hematopoietic stem cells, which can also turn into other immune cells, like T cells, based on their gene activity and environment. Pax5 plays a key role by controlling the expression of many genes. It guides B cell development and commitment to differentiate into mature B cells, the soldiers of our immune system. Within the blood cell-making system, B cells are the only ones to express Pax5.
Is Pax5 mostly involved in the development of B cells?
Not only. Pax5 is important to maintain the identity of B cells, too. Even if an early B cell is already committed to the B cell lineage, it still requires Pax5’s activity in order to maintain its course. If you remove Pax5, this cell can revert to an earlier, undifferentiated stage which again can give rise to other cell lineages of the hematopoietic system.
Pax5 modulates the expression of many genes for this to happen.
Yes, it’s a transcription factor that regulates the expression of hundreds of genes, both as an activator and a repressor. What we found in our latest study is that different protein domains within Pax5 play different roles in gene regulation.
Can you tell us more about these domains?
Although the function of Pax5 has been studied intensively and we’ve known about its basic functions since the 1990s, the mechanisms by which its domains regulate the expression of target genes in vivo remained poorly understood. Pax5 can be split up in the following domains: at one end of the protein, the ‘N-terminus’, there’s the paired domain, which is the DNA-binding domain. In the centre, there’s the octapeptide and the partial homeodomain. At the other end, the ‘C-terminus’, there’s the transactivation domain and the inhibitory domain.
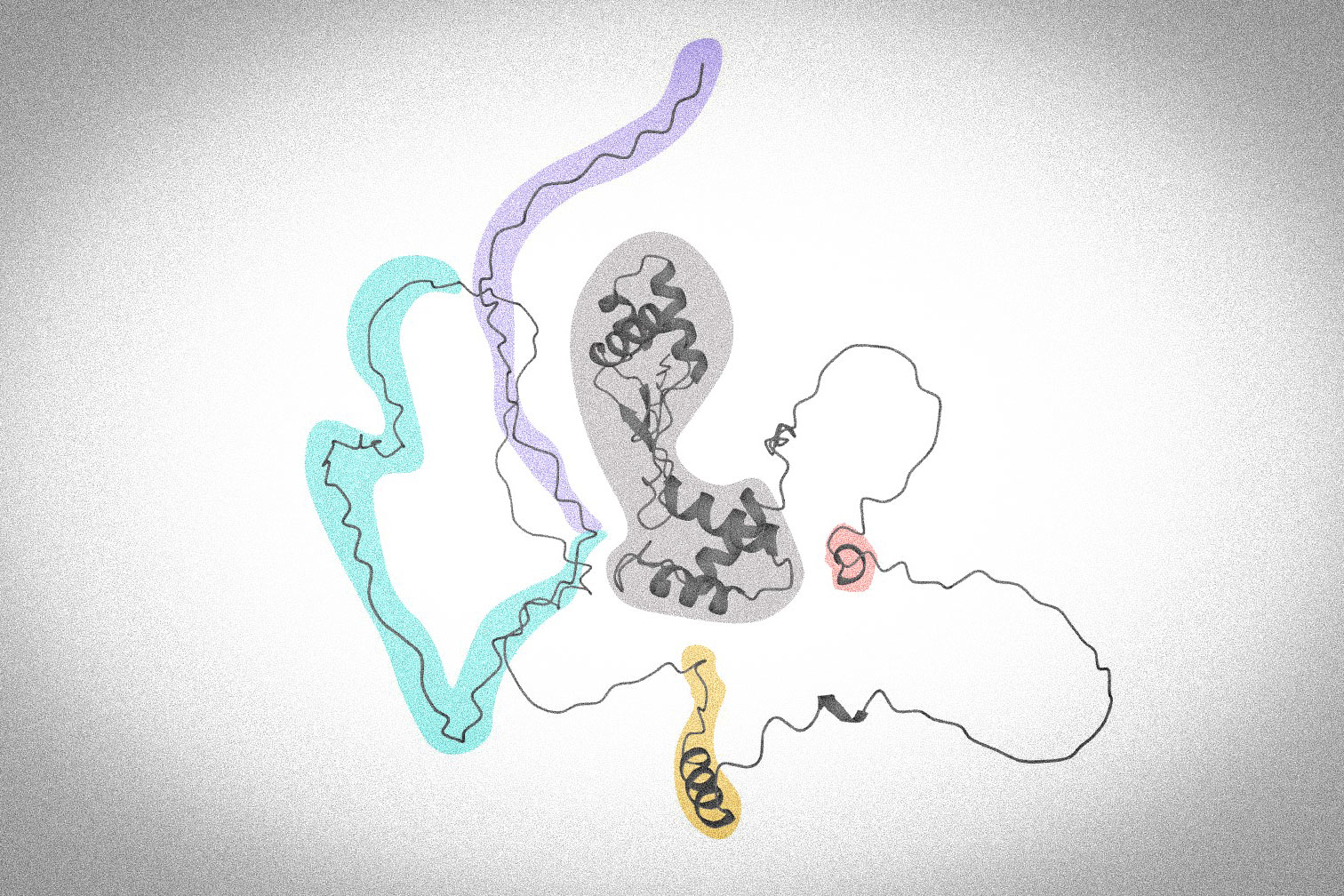
How did you investigate their roles?
It was important for us to gain insight into their function using mice as an in vivo approach. We artificially removed the DNA sequences that encode these domains from Pax5, which allowed us to observe the related consequences on gene expression in B cells. We found that the central domains have distinct functions: the partial homeodomain has mostly an activating role on target genes, while the octapeptide has more of a repressing role. They are necessary for optimal development in B cells. Our experiments show that the C-terminus domains are even more important for B cell development.
How so?
From our results, it seems that the two previously defined domains work as one and serve as the business end of Pax5. The more is removed from these domains, the more disturbed is Pax5’s function. In contrast to the previous functional separation of the two C-terminal domains based on in vitro experiments, our data shows that both C-terminal domains can activate and repress Pax5’s target genes in vivo. They recruit chromatin-modifying and histone-modifying complexes, which make Pax5-regulated genes accessible for the transcription machinery. The C-terminal domains are also required for ensuring that early B cells are committed to the B cell lineage and don’t differentiate into something else.
Could this research be directly relevant for medical applications?
Patients with acute lymphoblastic leukaemia often have mutations in the C-terminal domains of PAX5. Although our study was done in the mouse, we anticipate that the precise functions of PAX5’s domains in humans is similar. In my opinion, the beauty of basic research lies in finding answers to fundamental questions about how things work, which can be translated into new treatments. By understanding the function of Pax5 domains on a molecular level, we made a step in that direction.
You’re now done with your doctoral studies. What are you up to?
After my PhD, I wanted to continue my career in immunology, but in industry. Looking for jobs outside of the academic bubble was quite intimidating at first, but it was surprisingly easy for me to navigate job applications. I’m now using my expertise in immunology as a Medical Science Liaison at Janssen. I became interested in this job thanks to a career day that I attended in the context of the Vienna BioCenter PhD Symposium, organised by students. My work brings me closer to patients and healthcare professionals, which I’m very excited about.
Do you feel that the PhD programme prepared you for your job?
Absolutely. I often have to communicate with colleagues who don’t have a molecular biology background, for instance in sales or in healthcare. My PhD prepared me to be a confident presenter, and to be able to grasp complicated concepts very quickly. These skills are invaluable in my current position.
---
Original publication
Sarah Gruenbacher, Markus Jaritz, Louisa Hill, Markus Schäfer, Meinrad Busslinger: “Essential role of the Pax5 C-terminal domain in controlling B cell commitment and development”. Journal of Experimental Medicine (2023). DOI: 10.1084/jem.20230260
About the Vienna BioCenter PhD Program
Much of the work underlying this award was done by students in the Vienna BioCenter PhD Program. Are you interested in a world-class career in molecular biology? Find out more and apply in the current open call: https://training.vbc.ac.at/phd-program/
