Molecular Mechanisms of immune-evasion and therapy resistance
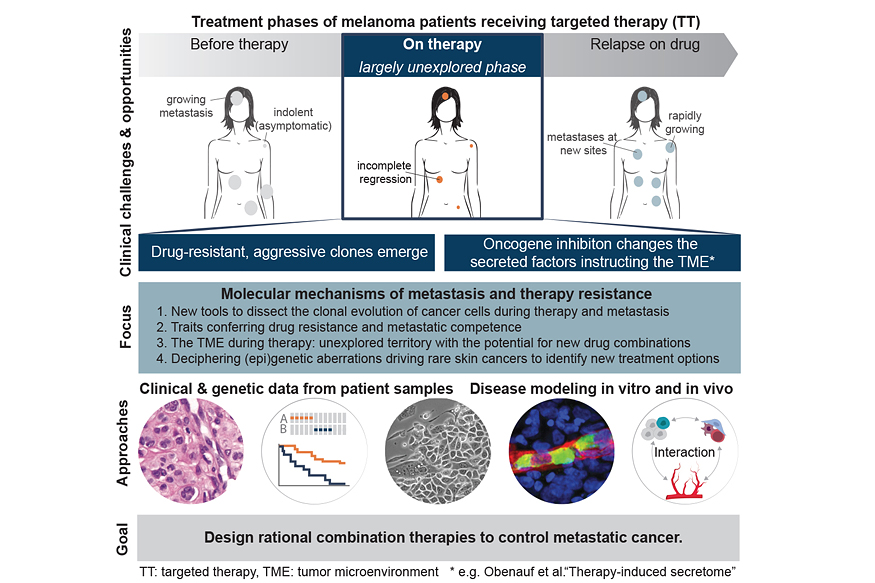
The treatment of metastatic cancer has undergone a paradigm shift in the last couple of years. The identification of specific ‘driver mutations’ in tumours holds the promise of tailored, mechanism-based treatment strategies, which are commonly referred to as ‘targeted therapies’. These new generations of targeted therapies can achieve tumour control for several months and have replaced unspecific cytotoxic chemotherapies for many cancer types. However, durable treatment responses are rare, due to the emergence of aggressive, drug-resistant clones that drive relapse and rapidly form new metastases (Figure 1). As a result, cure rates and long-term survival rates of metastatic patients treated with targeted therapies remain disappointingly low.
To better understand the clinical emergence of resistant cells, our work focuses on the poorly understood events during tumour regression. By combining the power of experimental model systems, in situ gene expression profiling techniques, and computational analysis, we recently discovered that targeted therapy with kinase inhibitors induces a complex network of secreted signals in drug-stressed melanoma and lung adenocarcinoma cells (Obenauf A et al., Nature, 2015).
This response, termed therapy-induced secretome (TIS), does not only enhance the survival of drug-sensitive cells, but also stimulates the proliferation, invasion, and metastasis of drug-resistant clones that are lurking in the background of the regressing tumours (Figure 2a-c). We also found that the regressing tumours in our animal models act as potent ‘magnets’ to attract drug-resistant cells from the circulation. This process, termed self-seeding, could add an additional layer of complexity to the treatment and relapse of patients on targeted therapy (Figure 2d). Our findings establish a general mechanism by which drug-stressed tumour cells can aggravate cancer progression and it could – at least partly – explain why targeted therapies rarely lead to complete tumour regression.
Drug-stressed cancer cells stimulate survival, proliferation, and metastasis of cancer cells
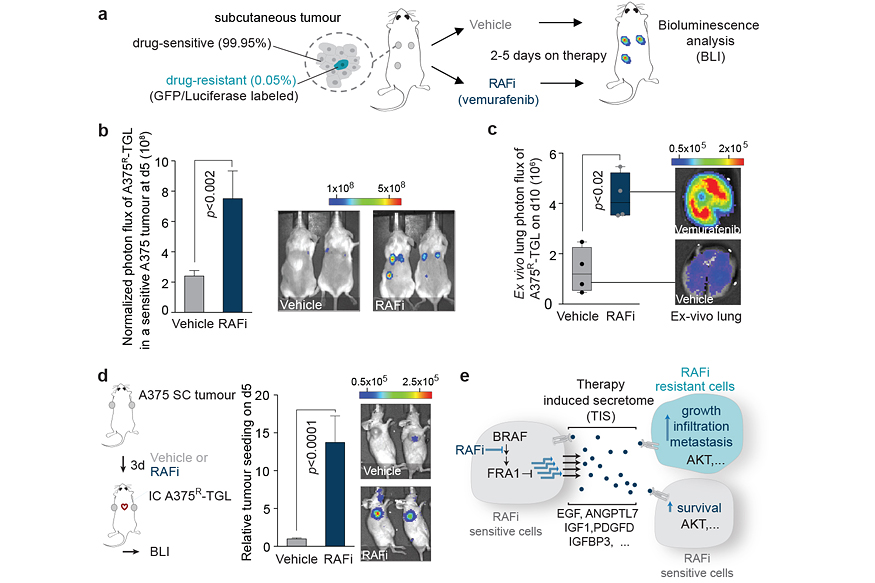
By dissecting the processes that lead to this phenotype in melanoma, we identified that the TIS consists of a vast number of signals that are capable of activating multiple signalling pathways, including an important survival and proliferation pathway (PI3K/AKT/mTOR pathway) (Figure 2e). The addition of AKT/PI3K/mTOR inhibitors blunted the outgrowth and metastasis of resistant cancer cells in animal models. These experiments suggest that this drug combination is a potential strategy to delay tumour relapse in patients. Even more importantly, our study started to expose the significant changes in tumours treated with targeted therapies. These changes are still largely unexplored and are expected to have a major influence on the efficacy of other drugs, such as immunotherapies, when given in combinations.
The research topics in our lab are centred around the mechanisms that drive disease progression and can be summarized in the following topics:
- New tools to dissect the clonal evolution of cancer cells during therapy and metastasis
- Traits conferring drug resistance and metastatic competence
- The tumour microenvironment during targeted therapy: unexplored territory with the potential for new drug combinations
- Deciphering genetic and epigenetic aberrations driving rare skin cancers to identify new treatment options
Selected Publications
-
Elewaut, A., Estivill, G., Bayerl, F., Castillon, L., Novatchkova, M., Pottendorfer, E., Hoffmann-Haas, L., Schönlein, M., Nguyen, TV., Lauss, M., Andreatta, F., Vulin, M., Krecioch, I., Bayerl, J., Pedde, AM., Fabre, N., Holstein, F., Cronin, SM., Rieser, S., Laniti, DD., Barras, D., Coukos, G., Quek, C., Bai, X., Muñoz I Ordoño, M., Wiesner, T., Zuber, J., Jönsson, G., Böttcher, JP., Vanharanta, S., Obenauf, AC. (2024) Cancer cells impair monocyte-mediated T cell stimulation to evade immunity. Nature.
-
Leiendecker, L., Neumann, T., Jung, PS., Cronin, SM., Steinacker, TL., Schleiffer, A., Schutzbier, M., Mechtler, K., Kervarrec, T., Laurent, E., Bachiri, K., Coyaud, E., Murali, R., Busam, KJ., Itzinger-Monshi, B., Kirnbauer, R., Cerroni, L., Calonje, E., Rütten, A., Stubenrauch, F., Griewank, KG., Wiesner, T., Obenauf, AC. (2023) Human Papillomavirus 42 Drives Digital Papillary Adenocarcinoma and Elicits a Germ Cell-like Program Conserved in HPV-Positive Cancers. Cancer Discov. 13(1):70-84
-
Haas L, Elewaut A, Gerard CL, Umkehrer C, Pedersen M, Leiendecker L, Krecioch I, Hoffmann D, Novatchkova M, Kuttke M, Neumann T, Pires de Silva I, Witthock H, Cuendet MA, Carotta S, Harrington KJ, Zuber J, Scolyer RA, Long GV, Wilmott JS, Michielin O, Vanharanta S, Wiesner T, Obenauf AC.(2021) Acquired resistance to targeted therapy confers cross-resistance to immunotherapy. Nature Cancer, 2, 693–708
-
Umkehrer C, Holstein F, Formenti L, Jude J, Froussios K, Neumann T, Cronin SM, Haas L, Lipp JJ, Burkard TR, Fellner M, Wiesner T, Zuber J, Obenauf AC. (2020) Isolating live cell clones from barcoded populations using CRISPRa-inducible reporters.
Nat Biotechnol. 2020 Jul 27 -
Obenauf, AC., Zou, Y., Ji, AL., Vanharanta, S., Shu, W., Shi, H., Kong, X., Bosenberg, MC., Wiesner, T., Rosen, N., Lo, RS., Massagué, J. (2015). Therapy-induced tumour secretomes promote resistance and tumour progression. Nature. 520(7547):368-72
Join us
- Master students and Post-docs: Contact Anna Obenauf with a letter of intent detailing why you want to join the lab.
- PhD students: Calls open 1 March and 1 September, apply here:
Vienna BioCenter PhD Program






